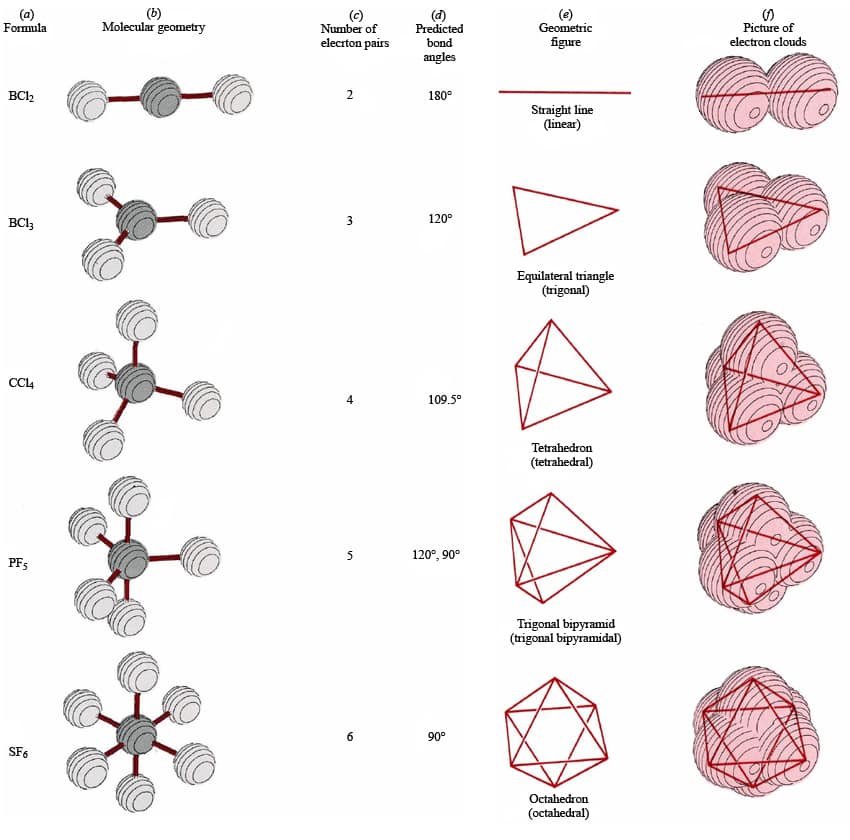
The regions of electron density will arrange themselves around the central atom so that they are as far apart from each other as possible. Use this number to determine the electron pair geometry.Įach bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. Step 3: Add these two numbers together to get the regions of electron density around the central atom. Make sure you understand why they are correct. The correct answers have been entered for you. How many lone electron pairs are on the central atom in each of the following Lewis structures? 
Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion. Crick.Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry).
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
